 The difference between them is that the electrostatic attraction holding giant structures together is stronger than the one holding simple structures. There are two types of structures that can contain covalent bonding: giant covalent structures and simple covalent structures. 
It's a strong electrostatic attraction between two positive nuclei and the shared pair of electrons between them. Moving on to Covalent Bonding, this type of bonding occurs between non-metals only. The negative ions of Oxygen are larger than the positive ions of Magnesium. Just like NaCl, Mg2+ ions and O2- ions are attracted to each other in its lattice, and they are also packed together in a cubic shape. It's pretty cool to think about how all these ions come together to form something we use in our everyday lives!Īnother example of a giant ionic lattice is Magnesium Oxide (MgO). The ions are packed together in a cubic shape, with the negative ions being larger than the positive ones. It's a giant ionic lattice where Na+ ions and Cl- ions attract each other in opposite directions. This type of compound can form giant ionic lattices, which are made up of a large number of the same ions repeating in a pattern.įor example, let's look at sodium chloride (NaCl). When this happens, strong electrostatic forces form between the oppositely charged ions, creating a lattice structure. Types Of Lattice Structures Giant Ionic Latticeĭo you remember learning about Ionic Bonding in our previous articles? Just to refresh your memory, this happens when electrons are transferred from metals to non-metals, causing metals to lose electrons and become positively charged ions (cations), while non-metals gain electrons and become negatively charged. Lattice structures are pretty fascinating when you think about all the different types and properties they can have. Covalent lattices can have varying qualities depending on the atoms involved. For example, ionic lattices are usually hard and brittle, while metallic lattices are malleable and ductile. And metallic lattices are made by the sharing of electrons between metal atoms.Įach type of lattice has its own unique properties. Covalent lattices happen when atoms share electrons. Ionic lattices are formed by the attraction between positive and negative ions. And that pattern is determined by the type of bonding happening between the particles. When we say “lattice,” we mean a three-dimensional pattern of ions or atoms in a crystal. Along the way, we’ll give some examples so you can visualize it better. And finally, we’ll look at the specific qualities of each one. After that, we’ll dive into the three types: ionic, covalent, and metallic. First, we’ll talk about what a lattice structure is. It all comes down to their chemical structures. And because of these differences, they also have different physical properties like how well they dissolve, when they melt, and how well they conduct electricity. But what does that even mean? Basically, each lattice has its own structure and bonding, which makes them different from each other. Ionic bonds are strong electrostatic forces between oppositely charged ions.If you’ve ever wondered how ionic, covalent, and metallic bonding are alike, here’s one thing: they can all make lattice structures. This is to keep things simple because ionic bonds can act in any direction. In three-dimensional models, ionic bonds are shown as straight lines between ions. The ionic lattice is held together by ionic bonds. A three-dimensional model for the ionic lattice in sodium chloride Ionic bonds This is why solid ionic compounds form crystals with regular shapes. The lattice arrangement continues in three dimensions. Remember that the lattice arrangement is giant - for example, a single grain of salt may contain 1.2 × 10 18 (1,200,000,000,000,000,000) ions. A two-dimensional model for the ionic lattice in sodium chloride 
The lattice is formed because the ions attract each other and form a regular pattern with oppositely charged ions next to each other. Instead, they have a regular, repeating arrangement called an ionic lattice. 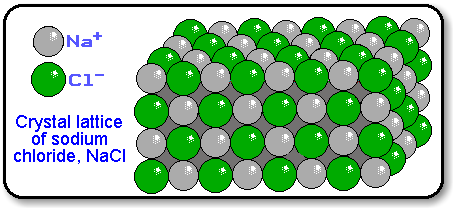 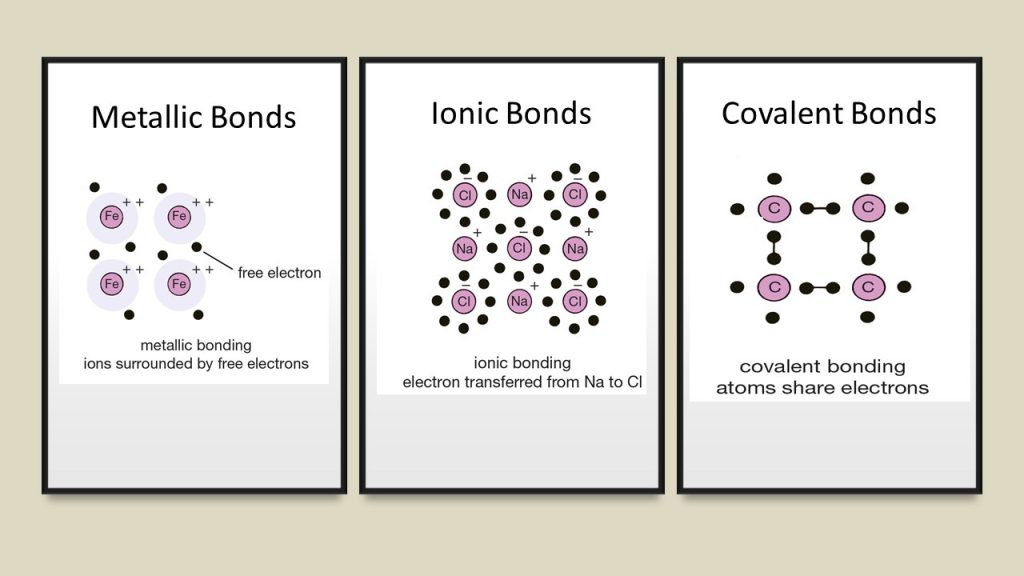
The ions in a solid ionic compound are not randomly arranged. The ionic lattice A regular arrangement of ions 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
